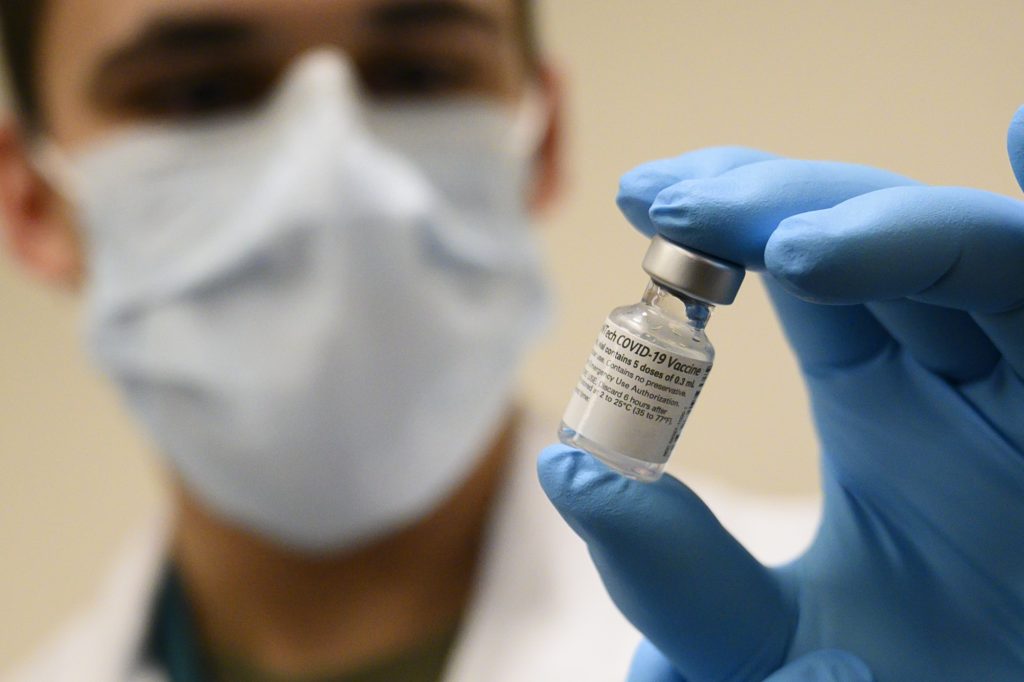
Children aged 12 to 15 can safely receive Pfizer’s Covid-19 vaccine, the United States’ Food and Drug Administration announced on Monday.
The expansion of vaccine availability to children in the US could bring the country “closer to returning to a sense of normalcy,” Dr. Janet Woodcock, the acting FDA commissioner, stated Monday.
Hoping to quell the fears of parents regarding the safety of inoculating their children, Dr. Woodcock stressed that the shot was put through a meticulous review by the FDA:
“Parents and guardians can rest assured that the agency undertook a rigorous and thorough review of all available data, as we have with all of our COVID-19 vaccine emergency use authorizations,” she assured the public.
Pfizer’s Covid-19 vaccine “100%” effective against the virus
The much-anticipated decision came after the pharmaceutical company applied for an emergency use authorization for its Covid-19 vaccine in adolescents aged 12 to 15 from the FDA in late March, citing data showing the shot’s safety and efficacy among children.
Pfizer conducted extensive clinical trials with participants in the age group, and found that its two-shot Covid-19 vaccine was incredibly effective in protecting them against the virus.
In its official announcement of the results, Pfizer stated that its vaccine against Covid-19 showed “100% efficacy” in preventing transmission of the virus, and produced a “robust antibody response” among those aged 12 to 15.
The study included 2,260 United States volunteers age 12 through 15, and it showed that there were zero cases of Covid-19 contracted among fully vaccinated children.

Trial results provide hope to parents of vulnerable children
The result was compared against the outcome of a control group receiving a placebo shot, in which 18 subjects developed the virus.
The news is extraordinarily welcome for those who have children suffering from a host of diseases, including cancer — who have waited with great difficulty for the shot to be approved for children.
Heather Ousley, the mother of a 13-year-old son who is battling a rare liver cancer, says that day cannot come soon enough, breaking into tears when she heard that he and his cohorts will most likely be able to begin receiving the precious vaccine soon.
“This day is the best day in the history of days!!! I love this day!!!” she texted as she joined other parents of ill children who have been praying for the time when the FDA would authorize the use of the inoculation.
Currently, Pfizer, Moderna, and Johnson & Johnson, the makers of the three coronavirus shots currently approved in the US, are conducting trials to determine the safety of the inoculation in young children.
Some research involves kids as young as six months.
Pfizer Covid-19 Vaccine very powerful against variants
Pfizer has disclosed that so far, its studies have shown that its shot is extraordinarily effective against the mutations that continue to circulate amongst the population.
The studies, which come out of the use of the vaccine after the spectacular successful rollouts in Qatar and Israel, suggest that the shot can indeed prevent the worst outcomes — including death — caused by B.1.1.7, the so-called British variant, as well as the South African variant, B.1.351.
Dr. Annelies Wilder-Smith, an infectious disease researcher at the London School of Hygiene and Tropical Medicine states to the New York Times “This is really good news. At this point in time, we can confidently say that we can use this vaccine, even in the presence of circulating variants of concern.”
It had been the working supposition that the vaccines did indeed fight the variants nearly as well as the original coronavirus strain, but that had yet to be scientifically proven.
According to the new studies, which were published in the New England Journal of Medicine, the promising conclusions were drawn from the clinical histories of more than 200,000 people who had received the inoculation in Qatar between February 1 and March 31.
Researchers found that the vaccine was between 87 to 89.5 percent effective at preventing infection with the British variant among those who were fully vaccinated.
In even more promising news, they found that it was nearly as effective with the more stubborn South African variant, preventing infection by as much as 72.1 to 75 percent in those who were fully vaccinated.
The vaccine was considered “highly effective” at protecting against not just death but severe pneumonia as well.
The extraordinarily positive findings showed that it was 97.4 percent effective at preventing severe, critical or fatal disease after the contraction of any form of the coronavirus. It was 100 percent effective at preventing severe, critical or fatal disease caused by either the British or the South African variant.
The British medical journal the Lancet published the results of the second study, which was conducted by Pfizer alongside researchers at Israel’s Ministry of Health. That study was based on more than 230,000 instances of coronavirus infection occurring in Israel between Jan. 24 and April 3 of this year.
At that time, the British variant was responsible for almost 95 percent of all coronavirus cases in the country, which by now has vaccinated more than half of its population.
The vaccine was more than 95 percent effective in protecting users against infection, hospitalization and death among all those who had been fully vaccinated. For those subjects who were 85 or older, the vaccine proved to be more than 94 percent effective at preventing infection, hospitalization and death.
See all the latest news from Greece and the world at Greekreporter.com. Contact our newsroom to report an update or send your story, photos and videos. Follow GR on Google News and subscribe here to our daily email!



