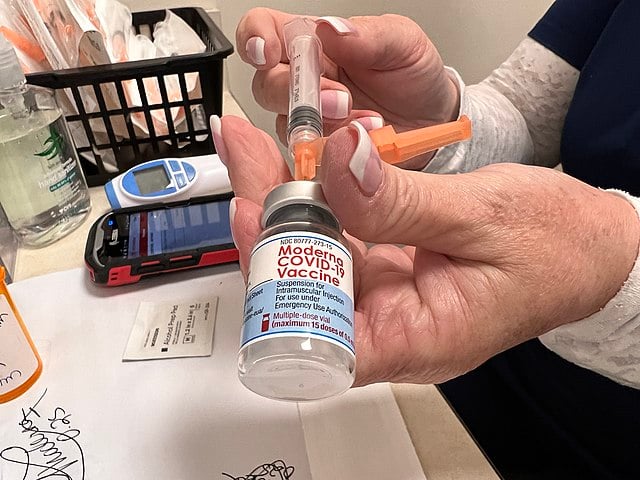
On Wednesday, the U.S. Food and Drug Administration (FDA) authorized two freshly revised COVID-19 boosters: one created by Moderna and one by Pfizer and German biotechnology company BioNTech.
The original SARS-CoV-2 coronavirus variant, which was the target of the first immunizations, as well as two omicron subvariants, known as the BA.4 and BA.5 lineages, are all protected against by both boosters.
In a statement, the FDA noted that according to STAT, BA.4 makes up the majority of the remaining cases whereas BA.5 accounts for roughly ninety percent of the current cases in the United States.
The upgraded Moderna booster can be administered to patients eighteen and older through an “emergency use authorization,” but the Pfizer-BioNTech booster can be administered to children as young as twelve years old.
People who had their last dose of their first COVID-19 vaccination series or their most recent booster shot at least two months ago may receive the single-dose vaccinations.
People Encouraged to Receive Booster Dose With Bivalent COVID-19 Vaccine
Dr. Robert M. Califf, the FDA commissioner, said in a statement on Wednesday that “as we head into fall and begin to spend more time indoors, we strongly encourage anyone who is eligible to consider receiving a booster dose with a bivalent COVID-19 vaccine to provide better protection against currently circulating variants.”
The new booster shots were approved without being put through official clinical testing in humans, much like the yearly influenza vaccination.
Instead, the FDA based its choice on a wealth of safety and effectiveness data acquired on the initial Moderna and Pfizer-BioNTech shots, which were launched in late 2020. The agency also took into account results from two recent clinical studies in which two vaccine makers used experimental boosters made to target BA.1, an older and no longer circulating omicron subvariant.
Lastly, the producers tested the most recent boosters, which target BA.4 and BA.5, on animals before the FDA approved the vaccines for use in people.
The director of the FDA’s Center for Biologics Evaluation and Research, Dr. Peter Marks, noted in the statement that “the FDA has extensive experience with strain changes for annual influenza vaccines,” and they are confident in their authorizations of these booster shots given the evidence.
Advocating for the Use of COVID-19 Boosters
The Centers for Disease Control and Prevention (CDC) must still advocate for the use of the boosters even if the FDA has already given its approval, according to STAT.
The vaccination advisory group for the CDC will convene on September 1st and September 2nd, and they might decide whether to recommend the booster shots as soon as today.
According to the FDA statement, “The agency will work quickly to evaluate future data and submissions to support authorization of bivalent COVID-19 boosters for additional age groups as we receive them.”
According to Pfizer-BioNTech and Moderna representatives, the booster dosages might be made available in two weeks if the CDC approves their broad usage. Similarly, Pfizer-BioNTech would only be available to those who were twelve years old or older whereas Moderna would only be available to those who were eighteen years old or older. If there is a difference in the protection offered by the vaccines, experts told The New York Times that it is still unknown what this difference might be.
See all the latest news from Greece and the world at Greekreporter.com. Contact our newsroom to report an update or send your story, photos and videos. Follow GR on Google News and subscribe here to our daily email!



