
A clinical trial has tested lab-grown blood on two people to observe how it performs inside the human body. It was the first time in the world for such a procedure.
Teams in Bristol, Cambridge, London and the NHS Blood and Transplant Department conducted studies in which tiny blood amounts equivalent to a couple of spoonfuls were tried out on the red blood cells that carry oxygen from the lungs to the rest of the body.
The ultimate goal for growing blood in a laboratory is to manufacture vital, but ultra-rare, blood groups that are hard to get hold of that are essential for people with blood disorders such as sickle cell and rare blood types.
Professor Cedric Ghevaert, the Chief Investigator and a specialist in Transfusion Medicine and Consultant Haematologist at the University of Cambridge and NHS Blood and Transplant, said: “We hope our lab-grown red blood cells will last longer than those that come from blood donors.”
He added, “If our trial, the first such in the world, is successful, it will mean that patients who currently require regular long-term blood transfusions will need fewer transfusions in the future, helping transform their care.”
Lab-grown red cells may last longer
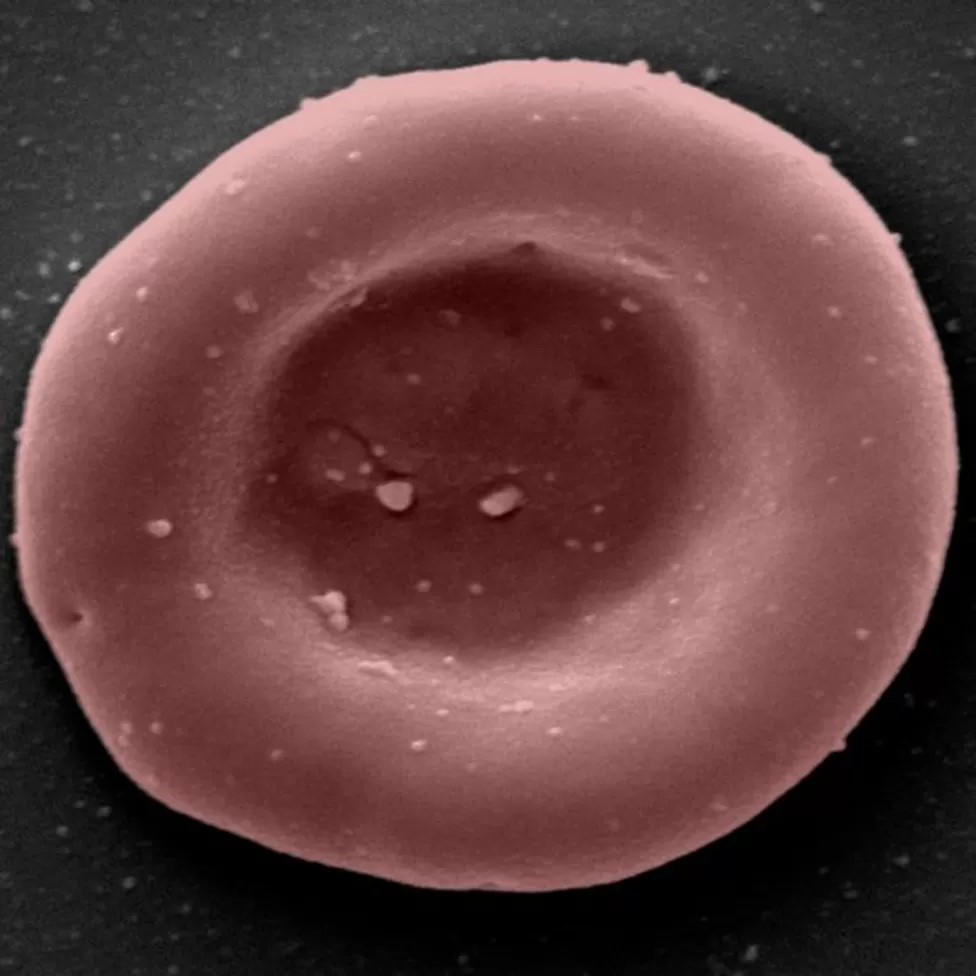
According to the research team, red blood cells normally last for around 120 days before they need to be replaced. Whereas a typical blood donation contains a mix of young and old red blood cells, lab-grown blood is all freshly made. For that reason, they believe that it may last a full 120 days.
Currently, two people have taken part in the trial. Yet the team aims to test the blood on at least 10 healthy volunteers. The participants will get two donations of 5-10mls at least four months apart: one of normal blood and one of lab-grown blood.
Manufactured blood has been tagged with a radioactive substance more often used in medical procedures. The reason is to enable scientists can see how long it lasts in the human body.
Researchers believe that the blood grown in the laboratory will be more powerful than normal. They also expect that it will allow for both smaller and less frequent donations in the future.
Identifying suitable blood from normal donors, in most cases, is difficult. For example, if the blood donated is not a precise match, then the recipient starts to reject it hence the treatment fails. As a result, the level of tissue-matching goes beyond the well-known A, B, AB and O blood groups. Prof Ashley Toye, from the University of Bristol, said some groups were “really, really rare” and there “might only be 10 people in the country” able to donate.
The process
The research team explained that to grow the blood, they start with a normal donation of a pint of blood (around 470ml) after, magnetic beads are used to fish out flexible stem cells that are capable of becoming red blood cells.
These stem cells are encouraged to grow in large numbers in the labs and then are guided to become red blood cells. The entire process that takes about three weeks and an initial pool of around half a million stem cells results in 50 billion red blood cells These are filtered down to get around 15 billion red blood cells that are at the right stage of development for transplant.
Professor Toye stressed that “We want to make as much blood as possible in the future, so the vision in my head is a room full of machines producing it continually from a normal blood donation.”
The challenges
Although the researchers are at the tip of realizing their anticipated results in the world’s first clinical trial, they face considerable financial and technological challenges.
Currently, the average blood donation costs the NHS around £130. However, for the researcher to grow the blood, it will cost massively higher at price unrevealed by the team.
In addition, harvested stem cells eventually exhaust themselves. This limits the amount of blood that can be grown, another justifiable challenge that will require more research to produce the volumes that would be needed clinically.
Dr. Farrukh Shah, the medical director of transfusion at NHS Blood and Transplant, said, “This world-leading research lays the groundwork for the manufacture of red blood cells that can safely be used to transfuse people with disorders like sickle cell,” adding “The potential for this work to benefit hard to transfuse patients is very significant.”
See all the latest news from Greece and the world at Greekreporter.com. Contact our newsroom to report an update or send your story, photos and videos. Follow GR on Google News and subscribe here to our daily email!



